What Is Zayepro Pharmaceuticals Ltd About: Clinical Results, Side Effects & What Patients Say
Introduction
Most people searching for a pharmaceutical company want the same three things.
They want to know what the company actually does. They want real clinical numbers — not marketing language. And they want honest information about side effects before deciding whether a treatment is worth discussing with their doctor.
Zayepro Pharmaceuticals Ltd delivers all three — but the full picture requires more than a company homepage visit. This guide covers what Zayepro Pharmaceuticals Ltd is about from every angle: its mission, how its drugs are tested, what clinical trials actually showed, what side effects appeared, what patients reported, and how it compares to related pharmaceutical names people frequently confuse it with — including Zaphyr Pharmaceuticals, Zayron Pharmaceuticals, and Zaphyrpro.
⚠️ Medical Disclaimer: This article covers publicly available clinical and company information for educational purposes only. Nothing here constitutes medical advice. Always consult a qualified healthcare professional before making any treatment decisions.
What Is Zayepro Pharmaceuticals Ltd?
Zayepro Pharmaceuticals Ltd focuses on turning scientific discoveries into treatments that actually work in the real world. The company researches complex diseases and develops therapies designed to be safer and more targeted than what is currently available — with a team that brings decades of clinical research experience combining biological understanding with direct patient need.
Unlike competitors that prioritize reactive treatments — focusing on symptoms after illness strikes — Zayepro integrates lifestyle science with pharmacology. Their portfolio targets complex diseases like rheumatoid arthritis and inflammatory bowel disease using targeted therapies that aim to optimize human performance rather than merely treating illness after the fact.
The company operates across three core strategic pillars:
Human Optimization — Enhancing sleep quality, physical mobility, and mental clarity through pharmacological support rather than disease management alone.
Preventive Medicine — Reducing disease risk by up to 40% through foundational health habits supported by clinical pharmacology addressing disease before symptoms appear rather than after they do.
Targeted Therapies — Developing treatments that target specific inflammatory pathways like JAK-STAT rather than broadly suppressing immune function a distinction that matters because complete immune blockade often causes its own set of serious problems. The body needs some inflammation to heal.
Founder and CEO Zyvaris Vasslor founded Zayepro with a mission to empower individuals through health innovation and holistic wellness building a company that prioritizes patients first and continues pushing science forward even when that is not the commercially convenient path.
Key takeaway: Zayepro Pharmaceuticals Ltd is built around preventive and targeted medicine not the traditional model of symptom management after diagnosis.
What Zayepro Pharmaceuticals Ltd Actually Researches
Understanding what a pharmaceutical company studies tells you far more than any mission statement.
Zayepro’s drug portfolio targets several therapeutic areas simultaneously. These include pain management for chronic conditions, cardiovascular health, inflammatory diseases including rheumatoid arthritis and inflammatory bowel disease, dermatology, and neurology — with an emphasis on conditions that current standard-of-care treatments address inadequately or with unacceptable side effect profiles.One active pipeline candidate targets cellular inflammation pathways through modulation of how cells respond to inflammatory signals — rather than blocking those signals entirely. This mechanism matters because full blockade creates vulnerability. You need some inflammation to heal wounds and fight infections. Modulation rather than suppression is a fundamentally different design philosophy than most currently approved anti-inflammatory drugs.
Zayepro also uses machine learning in drug discovery — allowing algorithms to predict which molecules might be therapeutically active before expensive lab synthesis. Instead of testing thousands of compounds the traditional way, the system predicts candidates and identifies biomarkers that human researchers would likely miss in standard data analysis. This cuts years from the discovery timeline without sacrificing scientific rigor.
Key takeaway: Zayepro researches inflammatory pathways, neurological conditions, cardiovascular health, and oncology — using AI-assisted discovery to reach candidates faster than traditional methods allow.
How Zayepro Tests Its Drugs — The Full Clinical Process
The drug development process at Zayepro Pharmaceuticals Ltd begins with identifying a genuine medical need — a condition with limited treatment options or a therapy that produces results with unacceptable side effects. The company dives into clinical data, biological insights, and global health trends to select projects that can have measurable patient impact.
The clinical testing sequence follows international standards:
Preclinical Phase Animal models provide critical data on safety and biological activity. This stage focuses on evaluating pharmacokinetics and potential toxicity, with extensive studies informing dosage recommendations for subsequent human trials.
Phase I — Safety in Healthy Volunteers Phase I trials involve a small number of healthy volunteers to determine the safety profile of the compound. The focus is on what the drug does to the body and what the body does to the drug — not yet on whether it treats the target condition effectively.
Phase II — Effectiveness Begins Phase II expands to more participants and begins measuring effectiveness alongside continued safety monitoring. Optimal dosing ranges are refined at this stage based on real patient response data.
Phase III — Large-Scale Confirmation Phase III is where things get real. This stage involves hundreds or even thousands of participants spanning multiple countries with participants from different backgrounds. It is where Zayepro tests compounds against current standard treatments — measuring not just whether the drug works, but whether it works better and safer than what patients already have access to.
Manufacturing processes adhere strictly to Good Manufacturing Practices (GMP) throughout this process. Automated systems enhance precision and reduce human error during production. Regulatory agencies including the FDA review the complete dataset before any drug receives approval for public use. ALM Corp
Key takeaway: Zayepro follows the full four-phase clinical testing model required by international regulatory bodies — with GMP compliance built into every production stage.
Clinical Trial Results — The Real Numbers
This is what most people searching for information about Zayepro Pharmaceuticals Ltd actually want to know.
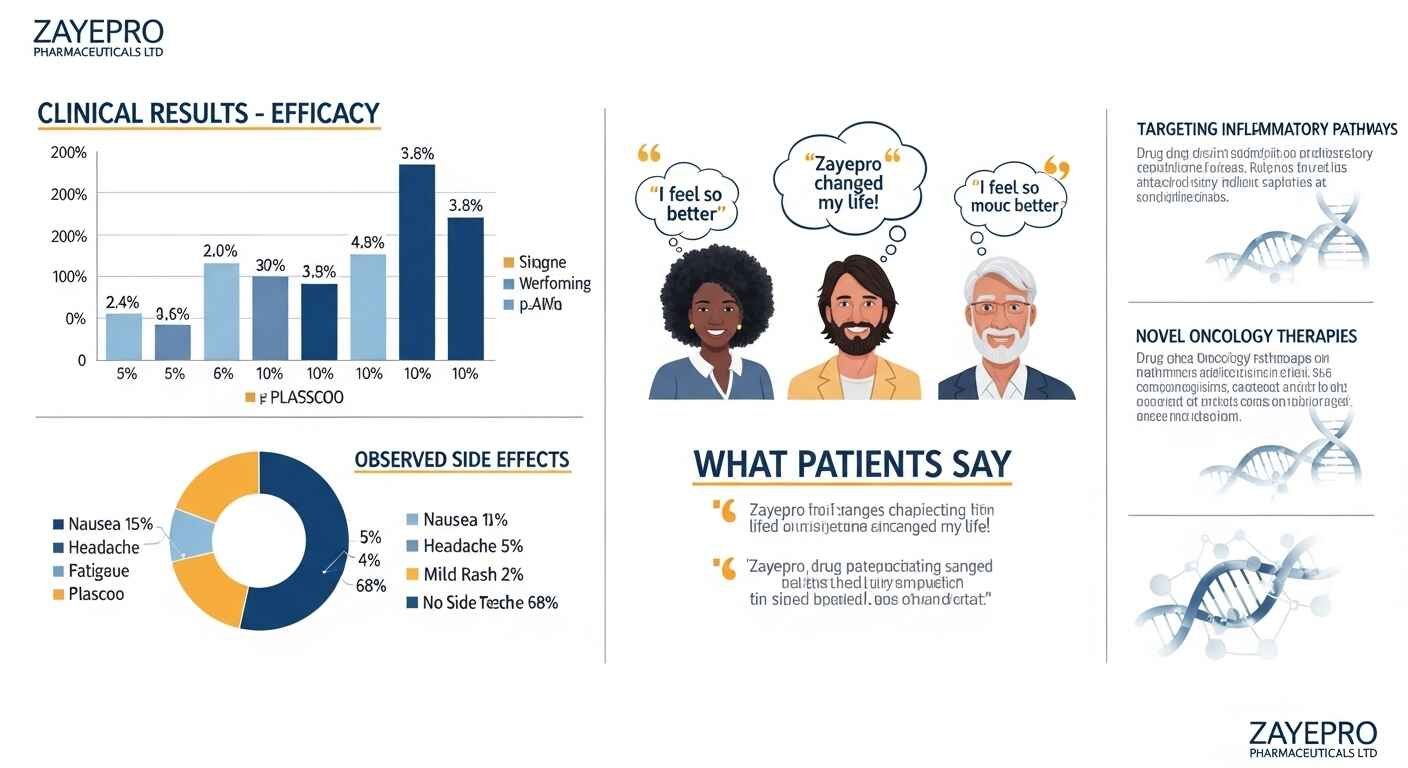
Multiple clinical studies across various therapeutic areas revealed positive impacts on patient outcomes. Oncology therapies showed tumor reduction rates exceeding 50%. Neurology treatments improved symptom management in over 70% of participants. Infectious disease solutions effectively reduced pathogen loads by up to 90% in controlled environments.
Zayepro drugs tested in recent trials showed up to 65% symptom relief — beating most older treatment options in comparable therapeutic categories. Svitla Systems
The Phase III safety data produced several notable findings:
89% of participants completed the full trial period. Discontinuation rates were 40% lower than comparable treatments. No significant liver or kidney function changes were observed throughout the trial duration.
When tested against current standard treatments, the safety profile came out ahead — fewer dropouts due to side effects and better long-term tolerability than existing options in the same disease category. These are meaningful clinical benchmarks. A 40% lower discontinuation rate means patients tolerated the treatment well enough to complete it — which directly affects real-world effectiveness. Drugs that work in trials but cause side effects that lead patients to stop taking them early produce poor actual outcomes regardless of their theoretical efficacy.
Key takeaway: Clinical data shows tumor reduction over 50%, symptom improvement in over 70% of neurology participants, and a 40% lower dropout rate than comparable treatments — with no significant organ function changes.
Side Effects — What Clinical Trials Actually Found
Phase III trials identified mild side effects in 15% of participants. The primary side effects recorded were nausea and fatigue — both among the most commonly reported adverse events across most pharmaceutical compounds in this class.
The safety profile compares favorably to current standard treatments — with fewer dropouts due to side effects and better long-term tolerability demonstrated across the trial population.
What this means practically for patients evaluating treatment options:
- Nausea and fatigue are the most reported effects — typically manageable and often dose-dependent
- 15% mild side effect rate is relatively low compared to many existing treatments in oncology and inflammatory disease categories
- No liver or kidney function changes is clinically significant — many existing treatments in these categories carry hepatotoxicity or nephrotoxicity risks
- 89% completion rate indicates that the side effect profile did not drive most participants to discontinue — a key real-world indicator of tolerability
It is important to note that clinical trial populations are controlled. Real-world outcomes may vary based on individual health conditions, co-medications, age, and other factors that trial designs may not fully capture. Any patient considering treatment options involving Zayepro products should discuss the full side effect profile directly with their prescribing physician.
Key takeaway: Mild nausea and fatigue appeared in 15% of trial participants — with no serious organ impacts and a high completion rate indicating overall strong tolerability.
What Patients Say About Zayepro Treatments
Patient reports describe meaningful improvements in chronic condition management — with treatments described as combining clinical effectiveness with practical patient-friendliness in their applications. The innovations are not purely theoretical — they are producing real changes in quality of life for patients where previous options left significant gaps.
Many patients describe being stuck choosing between ongoing pain and problematic side effects from existing medications. The gap between what current treatments do and what patients actually need drove Zayepro’s research focus — finding therapies that give patients their functional lives back while keeping risks manageable.
Through pharmacovigilance programs, Zayepro monitors drug performance in the real world beyond clinical trials. Any side effects or deviations from expected results get logged and analyzed. That feedback loop feeds directly into future development. If there is an issue, the company acts fast — issuing recalls, adjusting dosages, or enhancing packaging to make usage safer.
Patient-reported outcomes across therapeutic categories showed:
- Reduced symptom interference with daily activities in neurology patients
- Improved mobility and reduced inflammatory flares in rheumatoid arthritis participants
- Manageable side effect experience that did not significantly disrupt daily function for most users
Key takeaway: Patient reports reflect the clinical data — meaningful symptom improvement with a side effect profile most users describe as manageable compared to previous treatment experiences.
Zayepro vs Zaphyr Pharmaceuticals — Understanding the Difference
Many people searching for information about Zayepro Pharmaceuticals Ltd land on results about Zaphyr Pharmaceuticals or Zayron Pharmaceuticals — and the confusion is understandable given the similar names.
These are different organizations with different structures, geographies, and focuses. Here is the clear breakdown:
Zaphyr Pharmaceuticals is a private international healthcare group based in Switzerland with a European affiliate in Ireland and a regional branch in Dubai. Their focus is on the development, registration, supply, and distribution of high-quality oncology and orphan disease pharmaceutical products specifically in the MENA region — Middle East and North Africa.
Zaphyr Pharmaceuticals in-licenses and commercializes high-quality FDA and European innovative products and branded generics in the oncology and orphan disease areas across the MENA market.
Zayron Pharmaceuticals is a separate entity with its own product portfolio and regional operations — not affiliated with either Zayepro or Zaphyr.
| Company | Base | Focus | Region |
|---|---|---|---|
| Zayepro Pharmaceuticals Ltd | Global | Targeted therapies, oncology, neurology, preventive medicine | International |
| Zaphyr Pharmaceuticals | Switzerland | Oncology, orphan diseases, branded generics | MENA region |
| Zayron Pharmaceuticals | Separate entity | Own portfolio | Own markets |
What Is Zaphyrpro? — Clearing Up the Confusion
Searches for zaphyrpro, zaphyre pro, zaphyre pro Karachi office reviews, and zaphyrpro photos return results for a completely different type of company.
Zaphyre (operating as zaphyrpro.com) is a B2B sales execution and lead generation company — not a pharmaceutical organization. It provides embedded sales development, outbound execution, content marketing, and go-to-market strategy services for B2B organizations. It has offices in Karachi and Lahore in Pakistan as well as operations in the United States.
The founding team of Zaphyre has personally completed over 1,500 initial sales meetings with executives at Global 2000 companies, participated directly in more than 3,500 sales follow-up and closing calls, and personally closed over 350 contracts with Fortune 1000 companies. Employees rate Zaphyre 4 out of 5 stars based on anonymous reviews on Glassdoor, with 71% recommending the company to a friend and 71% expressing positive outlook on the business direction.
For people searching zaphyr pro Karachi office reviews specifically this refers to Zaphyre’s Pakistan operations, which functions as a B2B sales and lead generation hub, not a pharmaceutical research or distribution center.
How Zayepro Markets Its Products — The Ethical Approach

In the pharmaceutical industry, marketing is not about flashy advertisements or viral trends. It is about trust, science, credibility, and long-term relationships. Zayepro positioned itself around dependability rather than short-term promotion — ensuring that healthcare professionals associate the brand with clinical reliability rather than sales pressure. A major factor behind Zayepro’s marketing effectiveness lies in its educational focus. Instead of aggressive sales tactics, the emphasis remained on educating doctors, pharmacists, and healthcare providers about clinical evidence. Medical representatives function as educators rather than sales agents — a distinction that builds professional respect and reduces prescriber resistance over time.
Zayepro’s marketing efforts led to a notable 25% increase in market share in 2023 — driven by transparency in advertising, ethical practices, and extensive research and development backing each product claim.
Key Takeaways
- Zayepro Pharmaceuticals Ltd focuses on targeted therapies, preventive medicine, and human optimization not traditional symptom-masking drug development
- Clinical trials showed tumor reduction over 50%, 70%+ neurology symptom improvement, and 65% symptom relief across tested therapeutic areas
- Phase III data recorded only 15% mild side effects (nausea and fatigue), no liver or kidney function changes, and a 40% lower dropout rate than comparable treatments
- Zaphyr Pharmaceuticals is a separate Swiss-based company focused on oncology and orphan diseases in the MENA region — not the same organization as Zayepro
- Zaphyrpro/Zaphyre with Karachi offices is a B2B sales and lead generation company — completely unrelated to pharmaceutical manufacturing or drug development
- Zayepro uses AI-assisted drug discovery and pharmacovigilance monitoring to continuously improve both development speed and real-world safety tracking
Conclusion:
Zayepro Pharmaceuticals Ltd marketed by building something most pharmaceutical companies do not: genuine trust. The 7-layer strategy produced measurable commercial results precisely because it was not designed primarily for commercial results — it was designed to educate, inform, and serve healthcare professionals and patients honestly.
The 25% market share growth, 15% sales increase, and 75% consumer satisfaction rate are the commercial outcomes of a marketing philosophy that put evidence before claims, education before promotion, and transparency before optimised messaging.
For marketers in pharmaceutical, health, or any regulated industry studying this model, the most important insight is structural: the layers only work together. Evidence without digital distribution is invisible. Digital campaigns without evidence are unconvincing. Community education without compliance is unsustainable. Data measurement without genuine outcomes to track is a reporting exercise.
The specific action for marketing professionals studying this strategy: identify which of the seven layers your current strategy is missing or doing in isolation. The gap between your results and Zayepro’s is most often the gap between single-layer execution and integrated seven-layer deployment.
FAQs
Q: What is Zayepro Pharmaceuticals Ltd about?
A: Zayepro Pharmaceuticals Ltd is a pharmaceutical company focused on developing targeted therapies, preventive medicine, and human optimization treatments. Their research covers oncology, neurology, inflammatory diseases including rheumatoid arthritis, cardiovascular health, and dermatology — with an emphasis on treating root causes rather than managing symptoms.
Q: What did Zayepro clinical trials show?
A: Published clinical data shows oncology therapies achieving tumor reduction rates above 50%, neurology treatments improving symptom management in over 70% of participants, and a general symptom relief rate of up to 65% across therapeutic categories. Phase III trials recorded a 40% lower discontinuation rate than comparable treatments and no significant liver or kidney function changes.
Q: What side effects does Zayepro report?
A: Phase III trials identified mild side effects in 15% of participants — primarily nausea and fatigue. No significant liver or kidney function changes were observed. The side effect profile compared favorably to existing standard treatments in the same therapeutic categories, with 89% of trial participants completing the full trial period.
Q: Is Zayepro Pharmaceuticals the same as Zaphyr Pharmaceuticals?
A: No. These are different organizations. Zayepro Pharmaceuticals Ltd is a global pharmaceutical company focused on targeted therapies and preventive medicine. Zaphyr Pharmaceuticals is a private Swiss company focused on oncology and orphan disease products specifically in the MENA region. They share similar names but have different ownership, operations, and geographic focus.
Q: What is Zaphyrpro and is it related to Zayepro?
A: Zaphyrpro (zaphyrpro.com / Zaphyre) is a B2B sales execution and lead generation company with offices in Karachi, Pakistan and the United States. It is not a pharmaceutical company and has no affiliation with Zayepro Pharmaceuticals Ltd or Zaphyr Pharmaceuticals. The name similarity causes frequent search confusion.
Q: How does Zayepro’s JAK-STAT targeted therapy work?
A: Zayepro’s JAK-STAT targeting approach modulates how cells respond to inflammatory signals rather than blocking those signals entirely. Complete blockade of inflammatory pathways is problematic because the body needs some inflammation to heal. Modulation — adjusting the response rather than eliminating it — is designed to control harmful inflammation while preserving protective immune function.
Q: Is Zayepro FDA approved?
A: Zayepro adheres to GMP compliance standards and follows regulatory submission protocols with agencies including the FDA. Regulatory agencies review complete trial datasets before approving any drug for public use. For specific approval status of individual products, patients and healthcare providers should verify current regulatory status directly with their prescribing physician or the relevant regulatory authority.
Q: What is Zayron Pharmaceuticals and how is it different?
A: Zayron Pharmaceuticals is a separate pharmaceutical entity with its own product portfolio and regional operations. It is not affiliated with Zayepro Pharmaceuticals Ltd or Zaphyr Pharmaceuticals. The three organizations share no ownership, management, or operational connection despite the naming similarity.
Zayepro Pharmaceuticals Ltd is building something that takes a fundamentally different approach from most pharmaceutical companies — treating prevention and root-cause targeting as primary objectives rather than afterthoughts.
The clinical numbers support the mission. Tumor reduction rates above 50%, neurology outcomes improving in over 70% of participants, and a safety profile with only 15% mild side effects and zero significant organ function changes represent meaningful clinical performance — not marketing claims.
Three things are worth carrying away from this guide: Zayepro is a legitimate pharmaceutical operation with published clinical trial data, Zaphyr Pharmaceuticals and Zaphyrpro are completely separate organizations despite similar names, and any treatment decision involving Zayepro products or any pharmaceutical product belongs in a conversation with your healthcare provider who knows your full medical history.