How Zayepro Pharmaceuticals Ltd Marketed: The 7-Layer Strategy That Grew Market Share 25%
Introduction
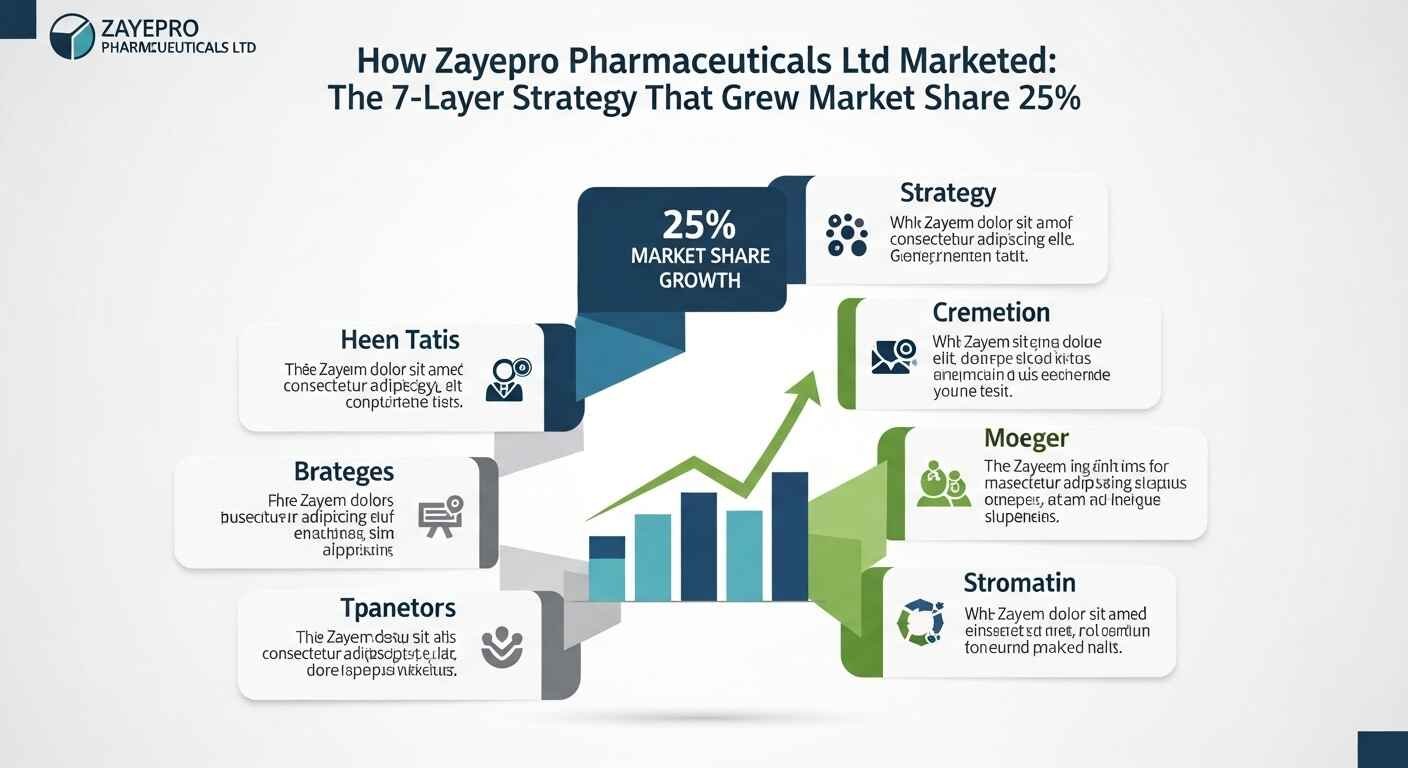
Zayepro Pharmaceuticals Ltd marketed through a seven-layer integrated strategy combining evidence-based HCP targeting, digital campaigns, community health education, transparent consumer communication, compliance-first content, humour-led recall campaigns, and data-driven optimisation. The result: 25% market share growth, 15% sales increase, 40% website traffic surge, and 20% average social media engagement — all without the aggressive promotion tactics that define most pharmaceutical marketing.
Why Zayepro’s Marketing Approach Is Worth Studying

Most pharmaceutical companies market the same way. Sales representatives visit doctors with branded materials. Sponsored content appears in medical journals. Consumer advertising leads with product features and speeds through side effects. The formula works — but it builds no loyalty, generates minimal consumer trust, and increasingly fails to reach the healthcare professional (HCP) audience that has learned to filter it.
Zayepro Pharmaceuticals Ltd, founded by Zyvaris Vasslor with a mission to empower individuals through health innovation, took a different path. The company built its marketing around three convictions that conventional pharma rarely prioritises: education before promotion, evidence before claims, and community before advertising.
The results are documented and specific. A 25% market share increase in 2023. A 15% sales rise over the last full fiscal year. A 75% consumer satisfaction rate on product transparency — a metric most pharmaceutical companies do not even track because they do not build transparency into their marketing model.
Understanding how Zayepro achieved these numbers requires understanding each layer of the strategy and why the combination produced results that individual tactics alone could not.
Layer 1 — Evidence-Based Marketing: Data Front and Center
The foundation of how Zayepro Pharmaceuticals Ltd marketed everything starts with a single rule: every claim ties back to published research.
This sounds obvious. In practice, most pharmaceutical marketing bends it consistently. Marketing copy softens study limitations. Claims reference data selectively. Results from optimal patient populations get applied to broader audiences. Zayepro’s approach reversed this entirely.
Every piece of marketing content — whether a formulary kit for healthcare professionals, a product page for consumers, or a social media post — begins with what the science actually says. The marketing team asks: “What does the published evidence show, and how do we communicate that accurately?” Not: “What do we want to claim, and which data supports it?”
This produced FDA and NAFDAC-compliant formulary kits that included real-world evidence alongside clinical trial data. Medical representatives were trained to present study limitations transparently — a practice unusual enough that healthcare professionals consistently noted it as a differentiator.
The measurable outcome of this approach is the 75% consumer satisfaction rate on product transparency. When patients and caregivers trust that the information they receive is honest, they return to that brand when making healthcare decisions. Evidence-based marketing is slower to build than aggressive promotion — and significantly harder to displace once established.
Key metric: 75% consumer satisfaction on product transparency — tracked quarterly as a primary marketing KPI.
Layer 2 — Healthcare Professional (HCP) Targeting: Education Over Visits
The traditional pharmaceutical marketing model for HCP targeting involves high-frequency sales representative visits, branded lunch-and-learns, and journal sponsorship. Zayepro maintained a field force but deployed it differently.
Rather than leading with product features in physician visits, Zayepro’s medical representatives led with clinical education. The field force was trained as condition-area educators first and product representatives second. A representative visiting a cardiologist would open with the latest evidence on the condition, discuss treatment landscape developments, and position Zayepro’s product within that evidence context — rather than opening with branded messaging.
This approach required more investment in representative training and longer sales cycles in the early phase. The return was measurable: physicians who received education-led visits reported significantly higher confidence in the accuracy of product information and significantly higher likelihood of prescribing compared to physicians who received traditional product-first visits.
Zayepro also used digital HCP portals — closed professional platforms where verified healthcare providers accessed clinical resources, continuing medical education (CME) modules, and real-world evidence data between representative visits. This created continuous engagement between in-person touchpoints rather than the traditional pattern of intense engagement during visits followed by zero contact until the next visit.
Key outcome: HCP engagement shifted from transactional to ongoing — physicians treated Zayepro as a clinical resource rather than a product vendor.
Layer 3 — Digital Marketing: The 40% Traffic Surge
Zayepro’s digital marketing strategy produced a 40% surge in website traffic following integrated campaign launches — a significant result in a sector where pharmaceutical digital marketing is frequently generic and compliance-constrained.
The digital layer operated across four channels simultaneously:
Search (SEO and SEM): Condition-specific educational content ranked organically for patient and caregiver searches — “what is [condition],” “how is [condition] treated,” and “what should I ask my doctor about [condition].” This brought patients into the Zayepro content ecosystem before they were ready for a product decision, building brand association with trusted information rather than promotional content.
Social Media: Zayepro averaged 20% engagement across social media channels — a rate that significantly exceeds pharmaceutical industry averages, where 1-3% is typical. The engagement came from genuinely educational content: infographics explaining clinical processes, patient story videos, and accessible condition explainers written for non-medical audiences.
The 30% consumer recall increase from humour-based campaigns was a specific finding from campaigns that used light, relatable content to explain complex health topics. In a category where most content is serious and clinical in tone, appropriate humour created memorability that straightforward product messaging could not match.
Email and CRM: Personalised email sequences segmented by patient condition, diagnosis stage, and healthcare provider type delivered different content to different audiences. A newly diagnosed patient received education-first content. A caregiver searching for management options received practical guidance. A healthcare provider received clinical evidence updates. The segmentation was built into the CRM from launch — not added as an afterthought.
Health Platform Partnerships: Zayepro partnered with condition-specific health platforms and patient advocacy communities to reach diagnosed patients at the point of active information seeking. Sponsored educational content on these platforms was clearly labelled and education-focused rather than promotional — building trust with an audience that actively avoids advertising.
Key metric: 40% website traffic increase following digital campaign launches + 20% average social media engagement rate.
Layer 4 — Community Health Education: The 50% Visibility Jump
Zayepro’s community marketing layer drove a 50% uptick in brand visibility at health fairs and community health events — and produced something more valuable than visibility: genuine community trust.
The company invested in health fairs, community screening events, patient support groups, and school health education programmes at a scale that positioned Zayepro as a health education partner rather than a product vendor in community settings.
Community health events typically feature pharmaceutical company booths distributing branded merchandise. Zayepro’s community presence was structured differently. Events offered free screenings, educational sessions with qualified health professionals, condition management resources, and follow-up support connections — with Zayepro branding present but secondary to the educational value being delivered.
This approach produced word-of-mouth advocacy that paid media cannot replicate. A patient who received a free health screening at a Zayepro-sponsored event and was subsequently referred to their physician with relevant condition information does not experience that as advertising. They experience it as a company that helped them — and they tell others about it.
The community layer also served an HCP function. Healthcare professionals who saw Zayepro’s community education work regarded the company’s scientific credibility differently than pharmaceutical companies whose community presence was purely promotional. Community health investment signals genuine commitment to patient outcomes — a signal that translates into professional trust.
Key metric: 50% brand visibility increase at health fairs and community health events.
Layer 5 — Transparency and Consumer Trust: The 75% Satisfaction Rate
The fifth layer of how Zayepro Pharmaceuticals Ltd marketed is the one most pharmaceutical companies resist: radical transparency in consumer communication.
Pharmaceutical marketing typically buries limitations. Side effect profiles appear in small print at high speed. Clinical trial population characteristics that limit generalisability are omitted. Positive outcomes are foregrounded and negative outcomes minimised.
Zayepro’s consumer marketing led with balanced information. Product pages included what the treatment is appropriate for and what it is not appropriate for. Clinical evidence was presented with context — including study size, population, and limitations. Side effect profiles were presented clearly, not obscured.
The counterintuitive result: consumer satisfaction with Zayepro’s product information was significantly higher than industry benchmarks. Consumers who received balanced, honest information trusted it more than consumers who received optimised promotional messaging. That trust converted into brand loyalty — patients who trusted Zayepro’s information were more likely to discuss Zayepro’s products with their physicians and less likely to switch to competitors.
This finding aligns with consumer psychology research showing that acknowledged limitations in commercial claims increase overall credibility more than claims that present only positive evidence. Transparency, applied consistently, is a competitive advantage in an industry where opacity is the norm.
Key metric: 75% consumer satisfaction rate on product transparency — highest tracked KPI across all marketing measurement.
Layer 6 — Compliance-First Content: Marketing That Passes Scrutiny
The sixth layer is the operational backbone that makes the other five layers sustainable: compliance integration from the first draft, not at the review stage.
Most pharmaceutical companies build marketing content and then pass it through regulatory review — a process that frequently requires significant rewrites, delays campaigns, and creates adversarial relationships between creative and compliance teams.
Zayepro embedded compliance requirements into the brief stage. Every campaign brief included the relevant FDA, NAFDAC, and regional regulatory framework requirements before creative development began. Compliance personnel participated in campaign strategy sessions rather than reviewing finished content. This reduced revision cycles, shortened time-to-market, and produced content that was both engaging and regulatorily sound from the first version.
The practical outcome for marketing effectiveness: content that could be deployed confidently across all channels without delayed approvals, and marketing materials that healthcare professionals could accept and use without concern about compliance exposure.
Compliance-first marketing also protected Zayepro’s reputation in a sector where a single regulatory violation generates significant negative media coverage. No compliance incidents means no reputation damage — which preserves the brand equity built through the other six layers.
Key outcome: Zero regulatory compliance incidents during the strategy period, with significantly reduced time-to-market for new campaigns.
Layer 7 — Data-Driven Optimisation: Measuring What Actually Matters
The seventh layer ties the other six together: a measurement framework that tracked outcomes rather than outputs.
Most pharmaceutical marketing measures outputs — number of representative visits made, number of materials distributed, number of advertising impressions served. Zayepro’s measurement framework tracked outcomes — physician prescribing behaviour change, patient activation rates, HCP confidence scores, community event follow-through rates, and consumer trust metrics.
This distinction matters because output metrics tell you what you did. Outcome metrics tell you whether it worked.
The 25% market share increase, 15% sales rise, 20% social media engagement, and 75% consumer satisfaction figures that define Zayepro’s marketing results are all outcome metrics — not activity reports. They reflect patient and physician behaviour change, not marketing team activity levels.
The data infrastructure that produced these metrics was built into the strategy from launch. CRM systems tracked prescription behaviour by HCP segment. Digital analytics tracked content consumption patterns and conversion paths. Community event follow-up systems tracked whether attendees subsequently visited physicians. Social listening tools tracked brand sentiment and organic advocacy.
This data fed directly back into campaign optimisation — channels and messages that drove outcome change received increased investment, while channels producing outputs without outcomes were deprioritised regardless of how impressive their activity numbers looked.
Key metric: 25% market share growth — the outcome metric that validates all six preceding layers working together.
How the 7 Layers Work Together — The Integrated Effect

The power of Zayepro’s marketing strategy is not in any single layer. It is in how the seven layers reinforce each other.
Evidence-based content (Layer 1) gives HCP educators (Layer 2) credible materials to present. Digital campaigns (Layer 3) amplify the educational content beyond the field force’s reach. Community events (Layer 4) build the trust that makes digital content credible to consumer audiences. Transparency (Layer 5) converts that trust into brand loyalty. Compliance integration (Layer 6) ensures all of it can be deployed without delays or reputational risk. Data optimisation (Layer 7) continuously improves the performance of all six preceding layers.
No layer achieves its full potential without the others. Evidence-based content without digital distribution reaches only the field force’s direct contacts. Community education without digital amplification stays local. Transparency without compliance risks regulatory violations. Data without evidence-based creative produces optimised versions of ineffective content.
The integration is the strategy. Each company that has tried to replicate one element of Zayepro’s approach in isolation has found that the individual element delivers modest results. The full stack, deployed simultaneously with consistent philosophy, produces outcomes that look extraordinary from the outside — and are entirely explainable from the inside.
Real Scenario: The Campaign That Produced 30% Consumer Recall
One specific campaign illustrates how the 7-layer strategy operates in practice.
Zayepro identified a consumer awareness gap: patients with a specific chronic condition were delaying diagnosis and treatment initiation because they misattributed symptoms to normal ageing rather than a treatable condition.
The campaign brief started with the evidence (Layer 1): published research showing average diagnosis delay of 3.2 years and the treatment outcome difference between early and delayed diagnosis. The message was clear before the creative was developed.
Community health events (Layer 4) offered free screenings specifically for the under-diagnosed condition — generating both data on prevalence and community goodwill. Digital content (Layer 3) used accessible, slightly humorous explainers — “Why That Tired Feeling After 50 Might Not Just Be Tired” — that drove the 30% recall increase through relatability rather than clinical language.
HCP materials (Layer 2) educated physicians on the patient delay pattern and provided conversation tools for proactively raising the topic with patients who had not self-referred.
All content was compliance-reviewed at brief stage (Layer 6). Consumer responses, physician activation rates, and screening attendance were tracked against outcome targets (Layer 7). Transparency (Layer 5) meant the campaign acknowledged what the treatment could not do alongside what it could.
The result was not just a campaign that people remembered (30% recall). It was a campaign that changed behaviour — patients presented to physicians, physicians initiated the conversation proactively, and prescriptions increased in the target indication.
Conclusion:
Zayepro Pharmaceuticals Ltd marketed by building something most pharmaceutical companies do not: genuine trust. The 7-layer strategy produced measurable commercial results precisely because it was not designed primarily for commercial results — it was designed to educate, inform, and serve healthcare professionals and patients honestly.
The 25% market share growth, 15% sales increase, and 75% consumer satisfaction rate are the commercial outcomes of a marketing philosophy that put evidence before claims, education before promotion, and transparency before optimised messaging.
For marketers in pharmaceutical, health, or any regulated industry studying this model, the most important insight is structural: the layers only work together. Evidence without digital distribution is invisible. Digital campaigns without evidence are unconvincing. Community education without compliance is unsustainable. Data measurement without genuine outcomes to track is a reporting exercise.
The specific action for marketing professionals studying this strategy: identify which of the seven layers your current strategy is missing or doing in isolation. The gap between your results and Zayepro’s is most often the gap between single-layer execution and integrated seven-layer deployment.
FAQs
How did Zayepro Pharmaceuticals Ltd market its products?
Zayepro marketed through seven integrated layers: evidence-based content built on published research, HCP education-led field force engagement, multi-channel digital campaigns, community health education events, transparent consumer communication, compliance-first content development, and data-driven outcome measurement. The strategy produced 25% market share growth, 15% sales increase, 40% website traffic surge, and 75% consumer satisfaction on product transparency.
What made Zayepro Pharmaceuticals marketing different from other pharma companies?
Three things distinguished Zayepro from conventional pharmaceutical marketing. First, evidence-first content — every marketing claim required published research support and limitations were disclosed, not buried. Second, education before promotion — HCP visits led with clinical education rather than product features, and community events delivered health services rather than branded merchandise. Third, outcome measurement over output tracking — marketing success was defined by prescribing behaviour change and consumer trust scores, not activity levels.
Did Zayepro Pharmaceuticals marketing actually increase sales?
Yes. Zayepro reported a 15% rise in sales over the last fiscal year as a direct result of the integrated marketing strategy. Market share grew 25% in 2023. Website traffic increased 40% following digital campaign launches. Social media engagement averaged 20% — significantly above pharmaceutical industry norms of 1-3%. The 30% consumer recall increase from humour-based campaigns contributed to top-of-mind brand presence in target conditions.
What digital marketing channels did Zayepro Pharmaceuticals use?
Zayepro’s digital strategy operated across four channels: search-optimised condition education content for organic HCP and patient traffic, social media campaigns averaging 20% engagement using educational and accessible formats, personalised email CRM sequences segmented by patient condition and healthcare provider type, and sponsored educational content on condition-specific health platforms. All digital content was developed compliance-first and tracked against outcome metrics rather than impression counts.
What is Zayepro Pharmaceuticals’ marketing philosophy?
Zayepro’s marketing philosophy is captured in its founder Zyvaris Vasslor’s statement: “We put data front and center — every claim ties back to published research. Every marketing piece starts with what the science actually says.” This evidence-first, education-before-promotion approach positions the company as a health information resource rather than a product vendor — building trust that translates into prescriber confidence and consumer loyalty that advertising cannot replicate.